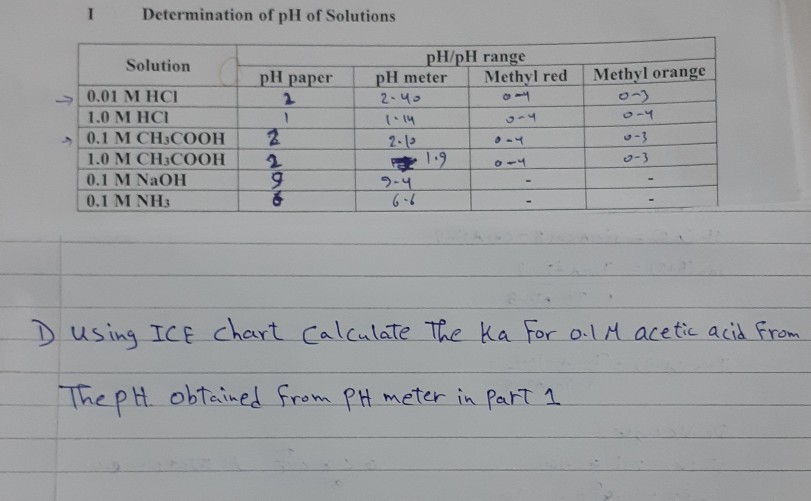
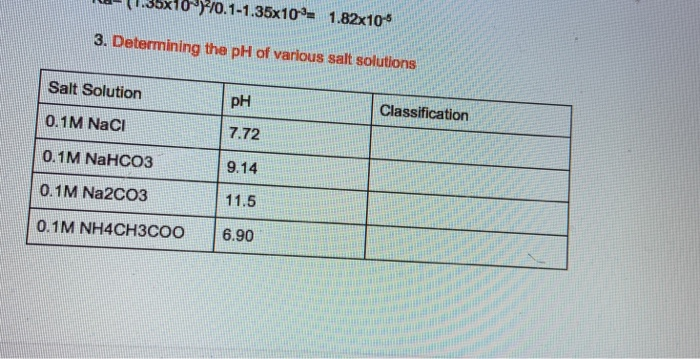
50 ml of 1 m ch3cooh solution when added to 50ml of 05 m naoh gives a solution with ph value 39 x 39 find the value of 39 x 39 pka of ch3cooh 48 owvxbegg -Chemistry - TopperLearning.com

SOLVED: A 40.0 ml sample of 0.01 M ethanoic acid (CH3COOH) is titrated with 0.02 M sodium hydroxide, NaOH. Calculate the pH after adding 30.0 ml of NaOH solution. *Given Ka of
pH of 0.1 M CH3COOH solution is 3 at 25 degree celcius . if limiting molar conductivity of CH3COO and H+ are 40 and 350 S cm2 mol . the molar conductance at 25 degree for 0.1M CH3COOH
![The pH of 0.1 M acetic acid solution is closest to[Dissociation constant of the acid, Ka = 1.8 × 10^-5 ] The pH of 0.1 M acetic acid solution is closest to[Dissociation constant of the acid, Ka = 1.8 × 10^-5 ]](https://haygot.s3.amazonaws.com/questions/1938773_1231298_ans_7f19b0e0d221405fbf38b0df680608aa.jpg)
The pH of 0.1 M acetic acid solution is closest to[Dissociation constant of the acid, Ka = 1.8 × 10^-5 ]
when 700ml of water is added buffer solution containing 0.01M CH3COOH and 0.1M CH3COONa then pH of final solution is(Ka of CH3COOH =1.8*10^ 5 ,log1.8=0.25)
![The P^(H) of 0.01 M solution of acetic acid is 5.0 What are the values of [H^(+)] and Ka respectively ? The P^(H) of 0.01 M solution of acetic acid is 5.0 What are the values of [H^(+)] and Ka respectively ?](https://d10lpgp6xz60nq.cloudfront.net/ss/web/577303.jpg)
The P^(H) of 0.01 M solution of acetic acid is 5.0 What are the values of [H^(+)] and Ka respectively ?

A buffer solution contains 100 mL of 0.01 M 700 mL of water is added pH before and after dilution are, if (pKa = 4.74) :
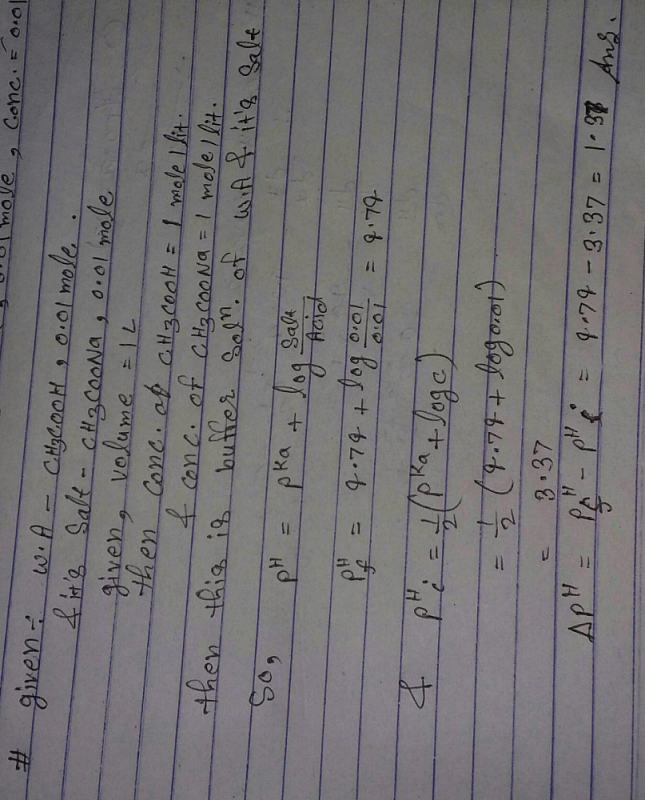






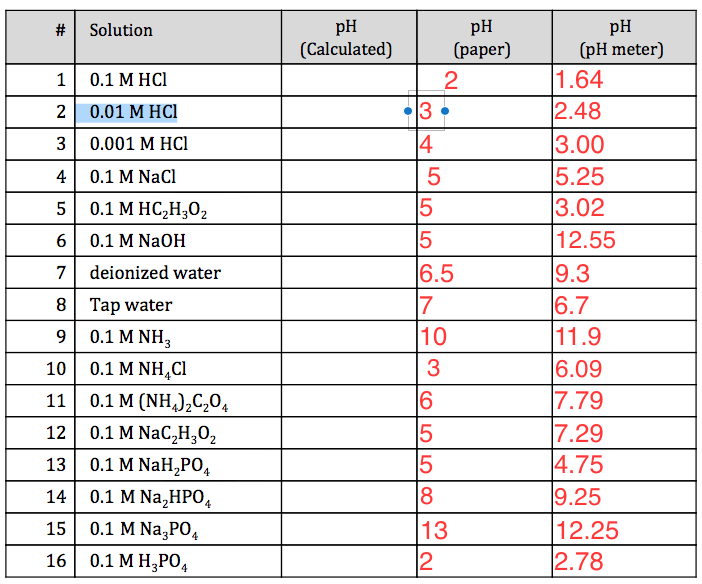

![The pH of 0.01 M solution of acetic acid is 5.0. What are the values of [H^+] and Ka respectively? The pH of 0.01 M solution of acetic acid is 5.0. What are the values of [H^+] and Ka respectively?](https://haygot.s3.amazonaws.com/questions/1445613_1156485_ans_e20f838d2ffe4d0eb21c5b51d09eb783.jpg)