
2.5 litre of 1 M NaOH solution mixed with another 3 litre of 0.5 M NaOH solution. Then find out molarity of resultant solution.
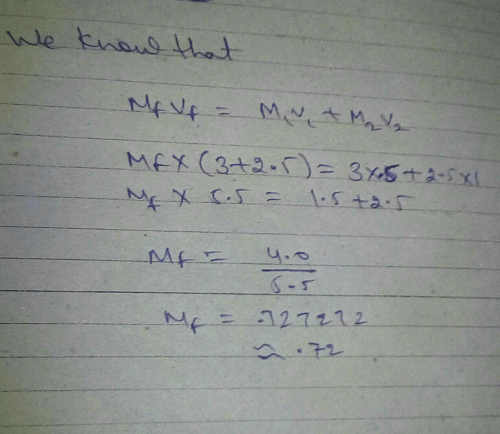
2.5 litre of 1M. NAOH solution mixed with another 3 litre of 0.5 NAOH solution then find out molarity of resultant solution? | EduRev NEET Question

LSV in 0.5 M NaOH solution containing of 20 mM glucose. The scan rate... | Download Scientific Diagram

LSV in 0.5 M NaOH solution containing of 20 mM of glucose. The scan... | Download Scientific Diagram

Preparation of 0.5 M EDTA Solution from Disodium EthyleneDiamineTetraacetate Dihydrate (EDTA.Na2.2H2O) - Laboratory Notes
Free Online Help: 50 ml of 0.5 M NaOH solution is diluted to 200 ml find the molarity of new solution ?

SOLVED: Preparation of 0.5 M NaOH solution by dilution Volume of NaOH stock solution (mL) 25.0m| 30 ^ B. Concentration of dilute NaOH solution (M) Calculation of theoretical molarity of the dilute

SOLVED: Calculate the weight of 97% solid sodium hydroxide required to prepare 50 mL of 0.5 M NaOH solution Calculate the volume of hydrochloric acid required to prepare 50 mL of 0.5

the volume of 05 molars naoh solution required for complete neutralization of 365 gram hcl is gi4mkkoo -Chemistry - TopperLearning.com

What volume of 0.2 M NaOH (in ml) solution should be mixed to 500 ml of 0.5 M NaOH solution - YouTube








:max_bytes(150000):strip_icc()/prepare-sodium-hydroxide-or-naoh-solution-608150_FINAL-696b52d6f90b4b1383ec8f95db73a1f3.png)



