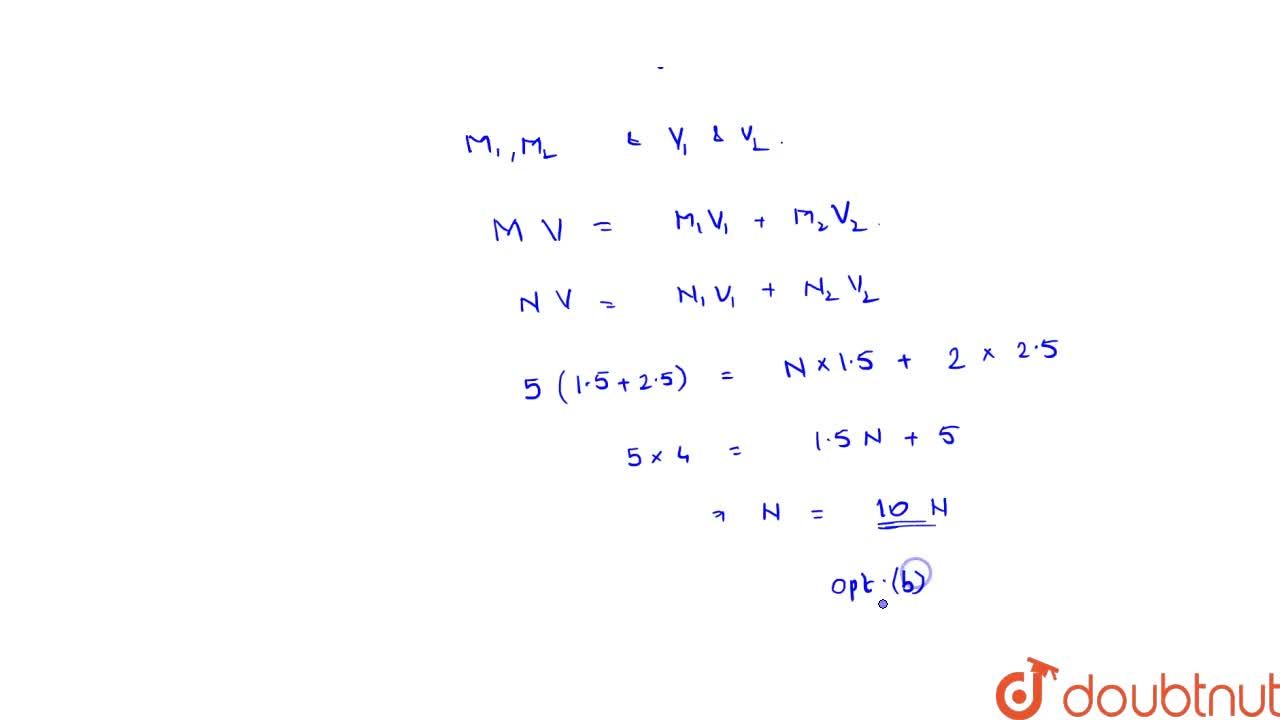
1.5 litre of a solution of normality N and 2.5 litre of 2 M HCl are mixed together. The resutant solution had a normality 5. The value of N is

1 M HCl and 2 M HCl are mixed in volume ratio of 4: 1. The resulting HCl solution is diluted to double its original volume. What is the final molarity of

Degree of Surface Coverage of Aluminium in 1.0M HCl and 1.5M HCl by HEC | Download Scientific Diagram

A drop ( 0.05 mL) of 12M HCl is spread over a thin sheet of aluminium foil (thickness 0.10 mm and density of Al = 2.70 g/mL). Assuming whole of the HCl
The volume of 1.5 M HCl required to completely react with 2.4 g Mg is - Sarthaks eConnect | Largest Online Education Community

Molar Mass & Molarity. Molar Mass Mass in grams of one mole of an element or compound. Numerically equal to the atomic weight of the element or the sum. - ppt download

A drop ( 0.05 mL) of 12M HCl is spread over a thin sheet of aluminium foil (thickness 0.10 mm and density of Al = 2.70 g/mL). Assuming whole of the HCl

SOLVED: How many mL of 1.5 M HCl solution is required to completely neutralize 20.00 mL of 10.0% calcium hydroxide solution (you can assume density is 1.00 g/mL), please show your work















