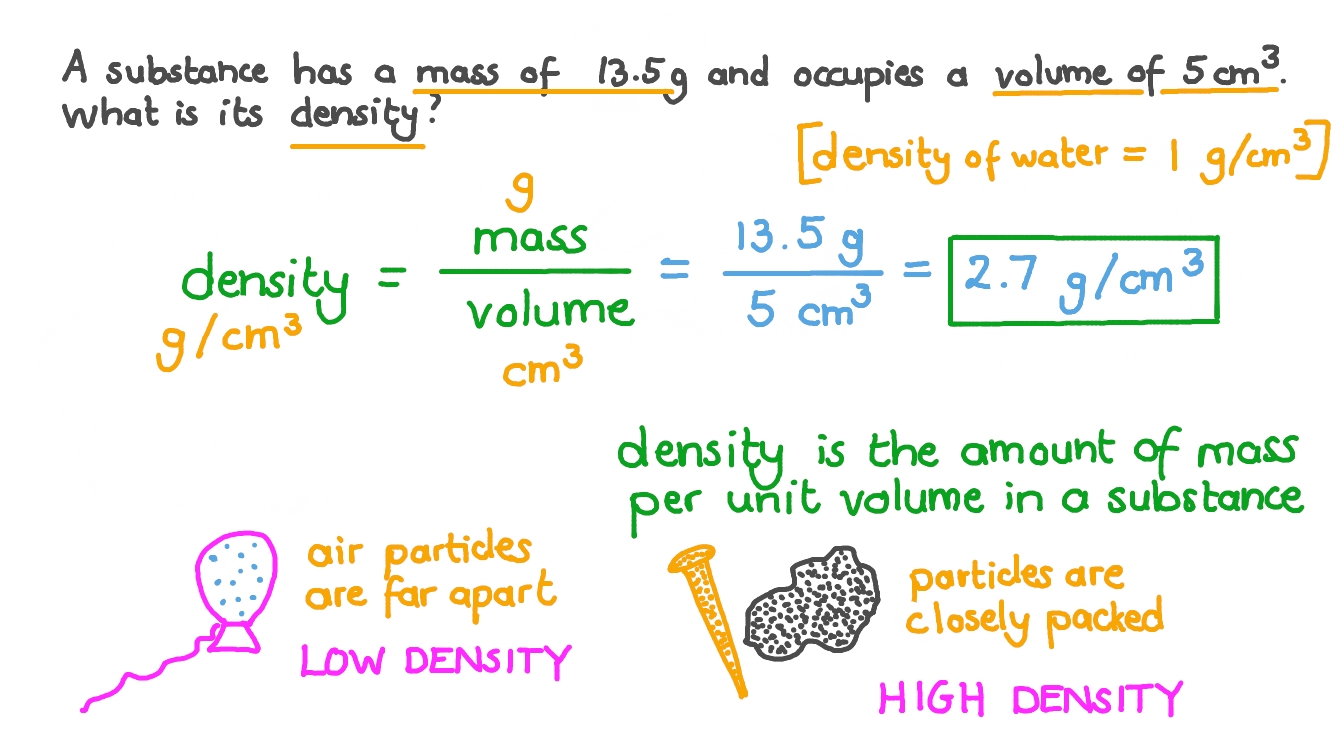The density of ice is 0.92 g cm^-3 and that of sea water is 1.025 g cm^-3 . Find the total volume of an ice berg which floats with its volume 800 cm^3 above water.

If the density of water is 1 g cm^(-3) then the volume occupied by one molecule of water is approximately

The density of water and ethanol at room temperature is 1.0 g / mL and 0.789 g / mL respectively. What volume of ethanol contains the same number of molecules as are

The density of ice is 0.92 g cm^-3 and that of sea water is 1.025 g cm^-3 . Find the total volume of an ice berg which floats with its volume 800 cm^3 above water.
The density of water is 1 g/cc. The numerical value of density of water in new unit kg/m^3 is Options: a) 1 b) 10 c) 10^2 d) 10^3

The density of water is 1.0g/cm^3. The density of the oil in the left column of the U-tube shown in the image is: a) 0.20 g/cm^3 b) 0.80 g/cm^3 c) 1.0g/cm^3 d)

The density of water is `1000 kg m^(-3)`. The density of water vapour at `100^() C` and 1 atmosp... - YouTube

Three litres of water (density 1gm/cm<sup>3</sup>) is added to twelve litres of methanol. (Density 0.8g/cm<sup>3</sup>). What is the density of the mixture?

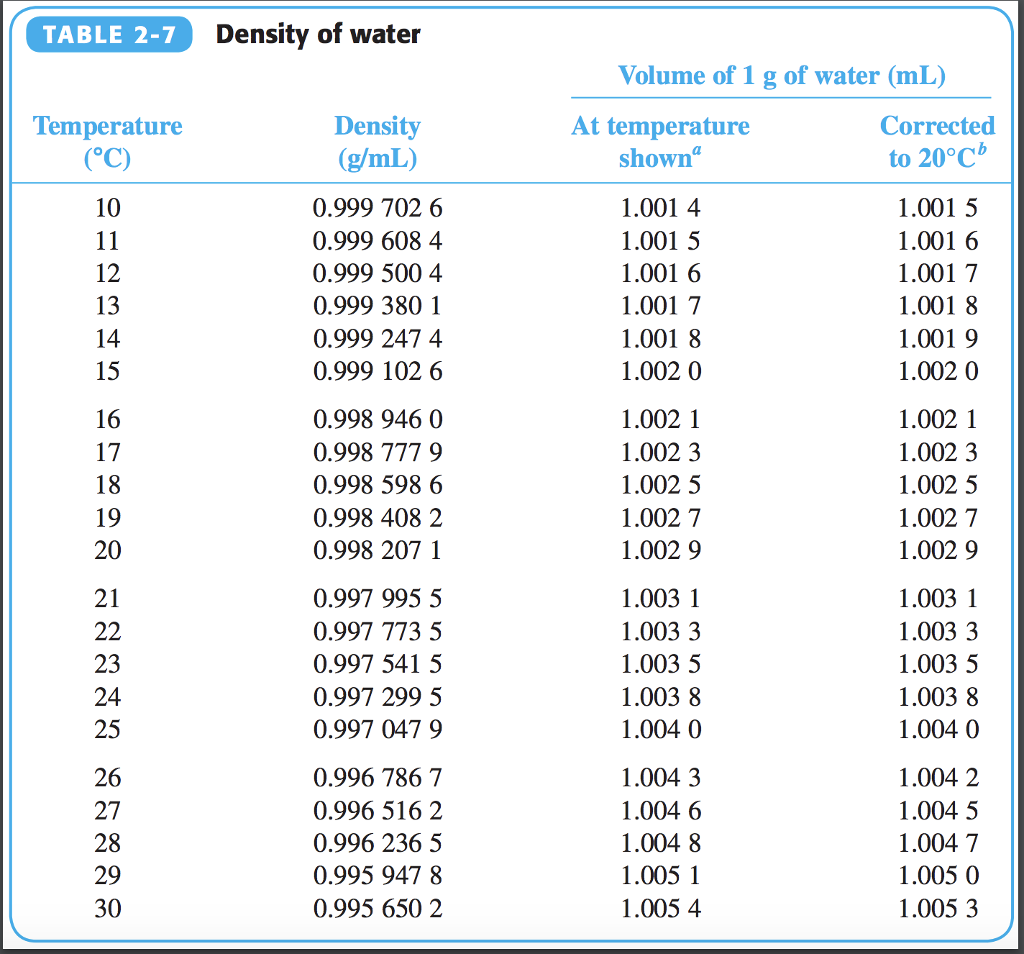
SOLVED: The density of water is well known t0 reach maximum at temperalure slightly above freezing- Table shown below from the Handbook of Chemistry and Physics (CRC Press 2009), glves experimental dala