
SOLVED: Calculate required HCL amount to prepare 1M HCL solution by using the given information below and prepare the solution. MWucr: 36,458 g/mole ducr: 1.19 gml 37% (wlv) stock concentration 2. Calculate

How much water should be added to `2M HCl` solution to form `1\"litre\"` of `0.5M HCl` ? . - YouTube

CHM256 - PREPARATION AND STANDARDIZATION OF HCl SOLUTION WITH PRIMARY STANDARD Na2CO3 SOLUTION - Studocu
500 ml of 0.2M BOH (a weak base) is mixed with 500 ml of 0.1 M HCl and the pH of the resulting solution is 9. What is the pH of a
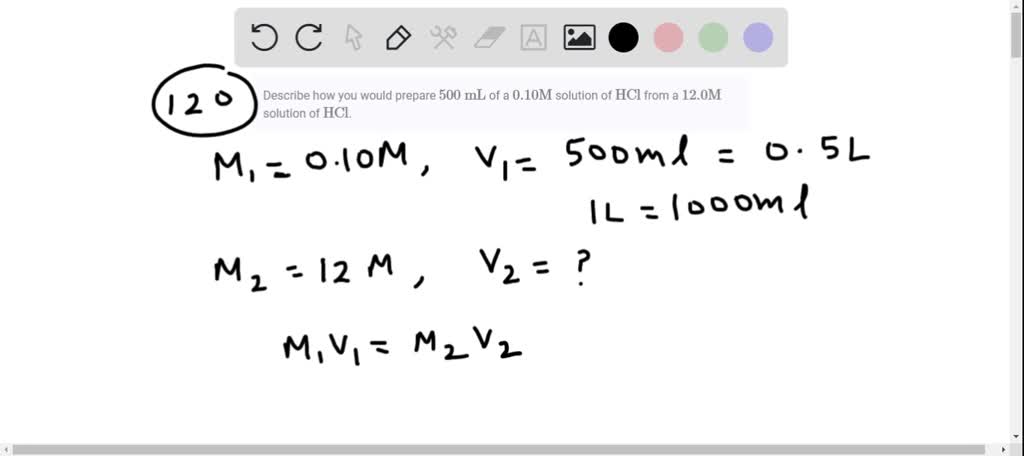

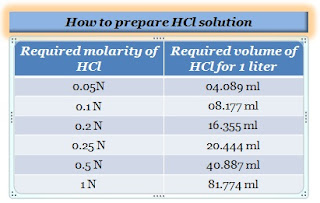


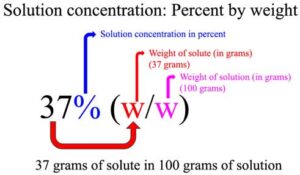






![Solved a) What is the [OH^-] of a 0.1 M HCl solution? b) | Chegg.com Solved a) What is the [OH^-] of a 0.1 M HCl solution? b) | Chegg.com](https://media.cheggcdn.com/media%2Fe3d%2Fe3de080a-4f47-4bae-b0f2-295c2277547f%2Fimage)




