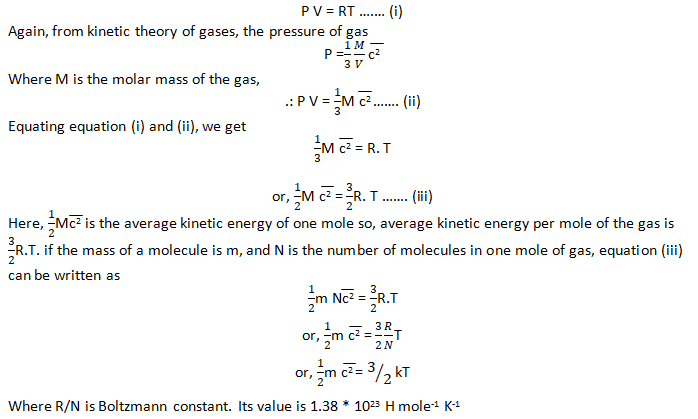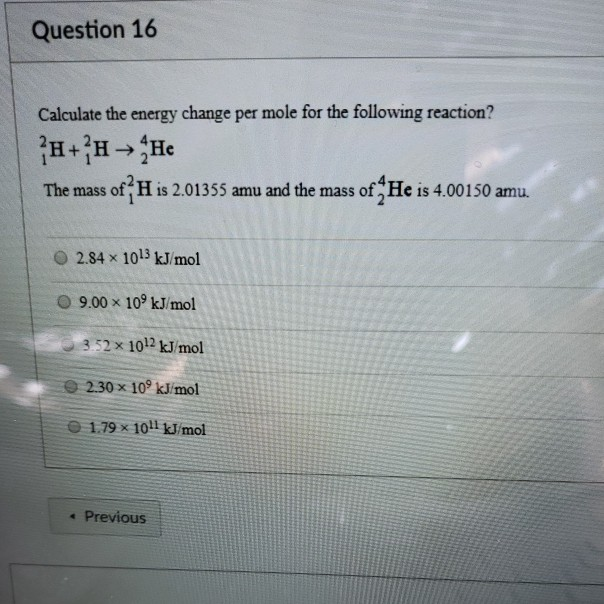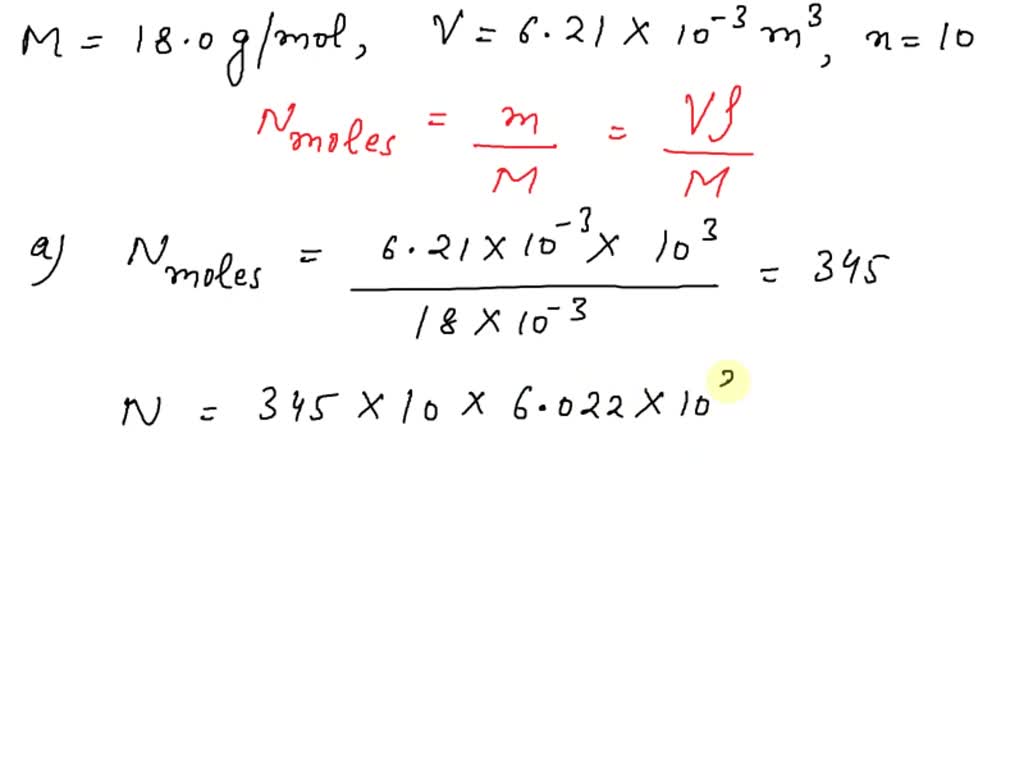
SOLVED: Water has a mass per mole of 18.0 g/mol, and each water molecule (H2O) has 10 electrons. (a) How many electrons are there in 6.21 liters of water? (1 liter is

Chemistry 1 Mole per Litre for Mole or Avogadro's Day " Greeting Card for Sale by Geek-topia | Redbubble

Is the textbook right ? If the unit of enthaply is KJ per moles why is it increasing if moles are increased? Shouldn't the increased increased energy be accounted by the increased